|
11/15/2023 0 Comments Full electron configuration of cobalt 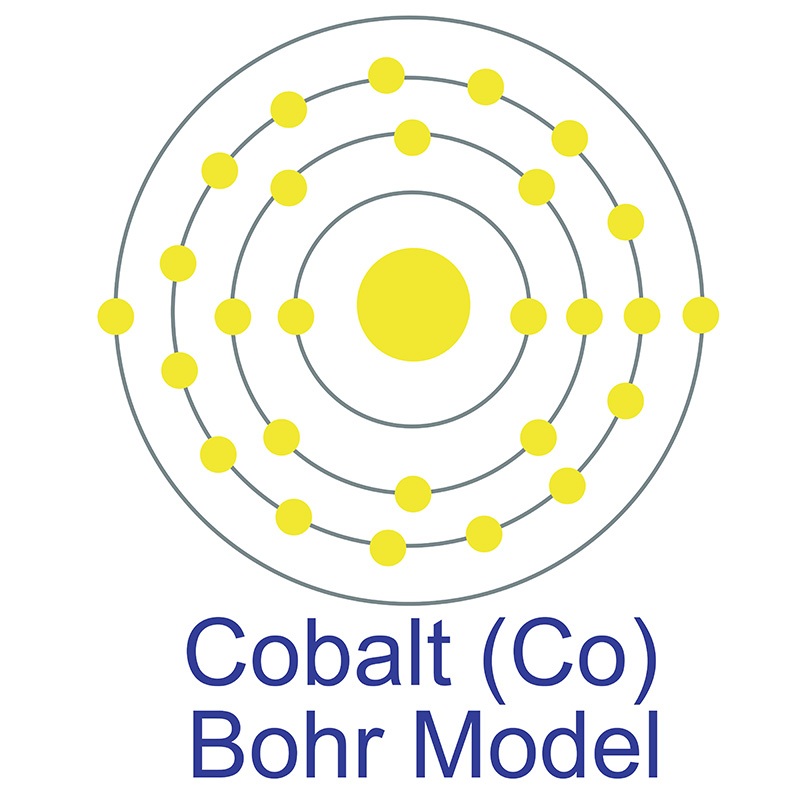
Home science Geography English medium Notes Hindi physics physical education maths english economicsĬhemistry business studies biology accountancy Political science History sociology Hindi sociology physics physical education maths english economics geography HistoryĬhemistry business studies biology accountancy political science Hindi Social science science Maths English Hindi social science science Maths English Hindi social science science maths English Ongoing research continues to explore new applications and advancements related to cobalt and its compounds while promoting responsible and sustainable practices in the cobalt industry. Its use in superalloys, lithium-ion batteries, magnets, healthcare, pigments, cutting tools, and nutrition highlight its importance. In summary, cobalt’s unique properties make it valuable in a range of industries. Responsible sourcing and sustainable practices are important considerations in the cobalt industry. While cobalt has valuable applications, it’s worth noting that cobalt mining and extraction can have environmental and social impacts. Cobalt deficiency can lead to health issues, and cobalt supplements or fortified foods are used to address deficiencies. It is a component of vitamin B12, which is necessary for various biological processes, including red blood cell formation and neurological function. These alloys exhibit excellent hardness, strength, and wear resistance, making them suitable for demanding applications.įurthermore, cobalt is an essential nutrient for humans and animals. Cobalt alloys, such as cobalt-chromium (Co-Cr) alloys, are used in the manufacturing of high-speed steel tools, cutting tools, and dental prosthetics. Additionally, cobalt-based dyes are used in the textile industry.Ĭobalt is a critical element in the production of cutting tools and wear-resistant materials. Cobalt salts are also used in the production of cobalt blue and cobalt green pigments. Cobalt blue, a vibrant blue pigment, is used in ceramics, glass, and artistic applications. This technique has been effective in treating certain types of cancer and is an important tool in oncology.Ĭobalt compounds have applications as pigments and dyes. 
Cobalt-60 emits high-energy gamma rays, which are used to target and destroy cancer cells in radiation therapy. In the field of healthcare, cobalt-60, a radioactive isotope of cobalt, is widely used in cancer treatment through a technique called cobalt therapy or cobalt-60 therapy. Cobalt magnets offer strong magnetic properties, high coercivity, and excellent temperature stability, making them valuable in applications such as electric motors, generators, and magnetic recording devices. It is a key component in the production of permanent magnets, particularly in the form of cobalt alloys. The high energy density and stability of cobalt-based cathodes contribute to the performance and longevity of lithium-ion batteries, which power numerous portable electronic devices, electric vehicles, and energy storage systems.Ĭobalt is utilized in the production of various magnetic materials. Cobalt compounds, such as lithium cobalt oxide (LiCoO2), are used in the cathodes of these batteries. These alloys are widely used in gas turbine engines for aircraft and power generation, where they can withstand extreme conditions and provide reliable performance.Ĭobalt is also a crucial component of lithium-ion batteries. Cobalt-based superalloys have exceptional strength, heat resistance, and corrosion resistance, making them ideal for high-temperature applications. One of the primary uses of cobalt is in the production of superalloys. Cobalt is known for its unique properties and finds applications in several industries. It is a transition metal that has a silvery-gray appearance. Some interesting facts of Cobalt are given below –Ĭobalt is the twenty-seventh element on the periodic table with the chemical symbol Co. Question : write some information about Cobalt ?Īnswer : Cobalt has melting point = 1,495Ĭobalt has density ‘8.9’ and it is found ”% on earth. Question : write the electron configuration of Cobalt element ?Īnswer : Cobalt electronic configuration is ” 3d7 4s2”. Answer : as we know Cobalt element is denoted by ‘Co’ symbol and Cobalt has ‘58.933’ atomic mass and ’27’ atomic number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
